A viral cruise ship outbreak triggered a wave of misinformation linking hantavirus to the COVID vaccine. The claim rests on a fundamental misreading of how pharmaceutical safety documents work.
1. What the Viral Claim Says
On May 7, 2026, a post on X by the account @TheProjectUnity read: “When you realise the new ‘Fear Virus’ being pushed by the media (Hantavirus) was also listed as an adverse side effect of the covid vaccines…”
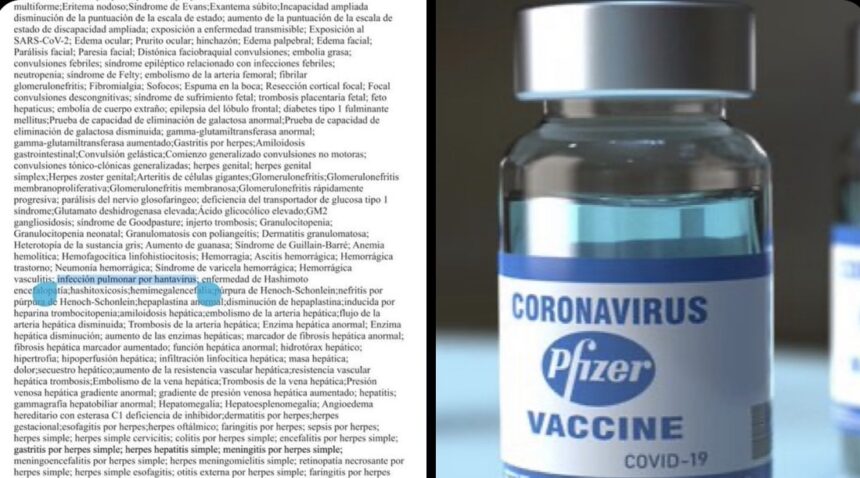
Separate posts on X and Instagram amplified the claim, with one widely shared Facebook post stating: “List of Pfizer Covid jab side effects includes Hantavirus pulmonary infection!” The posts shared a screenshot from a Pfizer document submitted to the U.S. Food and Drug Administration, with “hantavirus pulmonary infection” visibly highlighted on the page.
The implication, spread rapidly across social media platforms, was that Pfizer’s own documents confirmed the COVID vaccine caused hantavirus infection in recipients, and that the current cruise ship outbreak was, in effect, a vaccine injury event.
The claim was false.
2. What Actually Triggered This Rumor: The MV Hondius Outbreak {#the-mv-hondius-outbreak}
The timing of the claim is directly tied to a real and serious public health incident unfolding in real time.
In April 2026, an outbreak of hantavirus infection caused by the Andes virus was identified aboard the Dutch cruise ship MV Hondius, which had departed Ushuaia, Argentina on April 1. A passenger died from the virus on April 11. His body was taken ashore at Saint Helena on April 24, where his wife disembarked and died two days later in a Johannesburg hospital. A third passenger also died on board.
As of May 8, 2026, WHO confirmed eight cases in total, including three deaths, with a case fatality ratio of 38%. All six laboratory-confirmed cases were identified as the Andes strain of hantavirus.
As of May 12, 2026, the European Centre for Disease Prevention and Control reported 11 cases in total, nine confirmed and two probable, spanning passengers and crew from 23 countries including nine EU/EEA member states. The ship docked in Tenerife, Canary Islands on May 10, where disembarkation and repatriation of passengers was completed on May 11.
It was against this backdrop of genuine public alarm about a novel disease outbreak that the misinformation linking hantavirus to the COVID vaccine spread quickly and widely.
3. What Is the Pfizer Document People Are Citing? {#what-is-the-pfizer-document}
The document at the center of the claim is real. Understanding what it actually is, and what it is not, is the key to debunking this story.
The document, titled “5.3.6 Cumulative Analysis of Post-Authorization Adverse Event Reports of PF-07302048 (BNT162B2) Received Through 28-FEB-2021,” was approved on April 30, 2021, during the period when Pfizer’s vaccine was under emergency use authorization. It became public through a Freedom of Information Act lawsuit filed on September 16, 2021, by a group of more than 30 professors and scientists from universities including Harvard, Yale, Brown, and UCLA, who requested that the FDA share the data it relied upon in licensing Pfizer’s COVID vaccine.
The highlighted section showing “hantavirus pulmonary infection” appears on page 38 of the document, in Appendix 1, under a heading titled “List of Adverse Events of Special Interest.”
That heading is the critical distinction the viral posts ignored entirely.
4. What Is an Adverse Event of Special Interest? {#what-is-an-aesi}
This is where the misreading becomes clear.
Adverse events of special interest, or AESIs, are health conditions that researchers monitor closely during vaccine trials. They are not side effects. They are a pre-determined watchlist of medical conditions that scientists want to track to ensure that if any of them occur during or after a trial, they are flagged and investigated quickly.
The AESI list in the Pfizer document was compiled from multiple expert bodies and regulatory authorities, including the Brighton Collaboration, the European Medicines Agency ACCESS protocol, the U.S. Centers for Disease Control and Prevention, and the UK Medicines and Healthcare Products Regulatory Agency.
Hantavirus does not appear in the list of recorded adverse events that actually followed Pfizer’s COVID-19 vaccine rollout. Instead, it appears in the pre-specified list of events Pfizer committed to monitor. In other words, by adding hantavirus to this list, Pfizer said: if this condition ever appears in a vaccinated person, we will look at it more closely. At no point does the document suggest hantavirus was one of the adverse events that actually happened to a person who had received the vaccine.
Hantavirus is not mentioned in Pfizer’s product leaflet under the list of adverse events that were established to have a causal link to the vaccine.
A useful analogy: a hospital’s emergency room watchlist includes conditions like rabies, plague, and cholera. That does not mean the hospital is spreading those diseases. It means staff are trained to recognize and respond to them if they occur.
5. What Hantavirus Actually Is and How It Spreads {#what-hantavirus-actually-is}
Understanding the basic biology of hantavirus makes the claim even less credible.
Hantaviruses are a group of viruses carried by rodents such as mice and rats and transmitted through their droppings and urine. They can cause a range of diseases, from mild flu-like illness to severe respiratory illness.
The Andes virus is the only type of hantavirus known to spread person-to-person, and that transmission is usually limited to people who have close and prolonged contact with a symptomatic person, including exposure to saliva, respiratory secretions, or other bodily fluids.
There are no hantaviruses in the ingredient list of the Pfizer COVID vaccine, now called Comirnaty. According to Pfizer’s website, the Comirnaty shot does not contain live viruses of any kind.
A vaccine that contains no live virus cannot introduce, trigger, or cause an infection by a rodent-borne pathogen. The biological mechanism proposed by the claim simply does not exist.
6. What Pfizer, Regulators, and Scientists Have Said {#what-pfizer-and-regulators-said}
The response from authoritative sources has been consistent and unambiguous.
A spokesperson for Pfizer stated in an email that the listing of hantavirus pulmonary infection in the appendix of the document is not confirmation that the vaccine causes hantavirus pulmonary infection.
Independent fact-checkers at Snopes confirmed the document is authentic but found that hantavirus appeared not in a list of side effects, but in a pre-determined list of adverse events of special interest, which are unwanted medical events that occur after vaccination not necessarily caused by the vaccination.
Full Fact, the UK-based independent fact-checking organization, rated the claim false outright. Snopes and Lead Stories reached identical conclusions independently.
7. The Large-Scale Safety Data on mRNA COVID Vaccines {#large-scale-safety-data}
Beyond the document, the broader body of post-authorization vaccine safety research further undermines the claim.
Multiple large-scale studies have confirmed the safety of COVID-19 mRNA vaccines. One study published in JAMA Network Open in 2025 examined 28 million people in France and found no evidence linking COVID mRNA vaccination to hantavirus or any new infectious disease.
The Vaccine Adverse Event Reporting System, or VAERS, which is jointly run by the CDC and the FDA in the United States, is frequently misused in vaccine misinformation. Anyone with internet access can submit a report to VAERS, and the system itself carries an explicit public warning that a listing in VAERS does not constitute evidence of a causal relationship between a vaccine and any reported health event.
The VAERS warning exists precisely because the kind of reasoning behind the hantavirus-vaccine claim, “this condition appears in a document connected to the vaccine, therefore the vaccine causes this condition,” is the specific logical error the system is designed to prevent.
8. Verdict {#verdict}
| Claim | Verdict | Reason |
|---|---|---|
| Pfizer’s document lists hantavirus as a side effect | False | The document lists it as an Adverse Event of Special Interest, not a confirmed side effect |
| The COVID vaccine can cause hantavirus | False | Hantavirus is rodent-borne; the vaccine contains no live virus |
| The cruise ship outbreak is linked to COVID vaccines | False | The outbreak is caused by the Andes strain, transmitted through close contact with infected individuals |
| The Pfizer document is fake | False | The document is authentic, but has been misread |
The misinformation works by exploiting a gap between how pharmaceutical safety documents are structured and how most people naturally read them. A condition appearing on a monitoring watchlist reads, at first glance, like a condition the manufacturer is admitting the product causes. It is not. The distinction matters enormously, and the fact that multiple independent fact-checking organizations across different countries reached the same conclusion within days of the claim spreading is itself a signal worth noting.
9. Related Coverage on BrieflyUSA {#related-coverage}
This fact check is part of BrieflyUSA’s ongoing coverage of major public health and geopolitical developments this week. For related reporting, see:
- Mayo Clinic’s AI Detects Pancreatic Cancer Three Years Early — The breakthrough from BrieflyUSA’s science desk on what may be the most significant early cancer detection development in a generation.
- Trump Calls for Obama’s Arrest in Overnight Social Media Outburst — Our report on the overnight Truth Social posts and the DOJ grand jury opened in connection with DNI Gabbard’s declassified documents.
- Iran Considers Armed Dolphins and Cable Sabotage in Hormuz Standoff — BrieflyUSA’s coverage of the unconventional escalation options reportedly on Tehran’s table as the Strait of Hormuz standoff deepens.
- Mamdani Calls for Return of Koh-i-Noor During King Charles Visit — The New York mayor’s remarks during the royal visit and what they reveal about his political priorities.
This report draws on verified fact-checks published by Reuters, Snopes, Full Fact, and Lead Stories, the original Pfizer FDA submission document released under FOIA, and outbreak data published by the WHO, CDC, and the European Centre for Disease Prevention and Control. BrieflyUSA does not independently verify pharmaceutical regulatory filings but reports on conclusions reached by multiple independent fact-checking organizations.