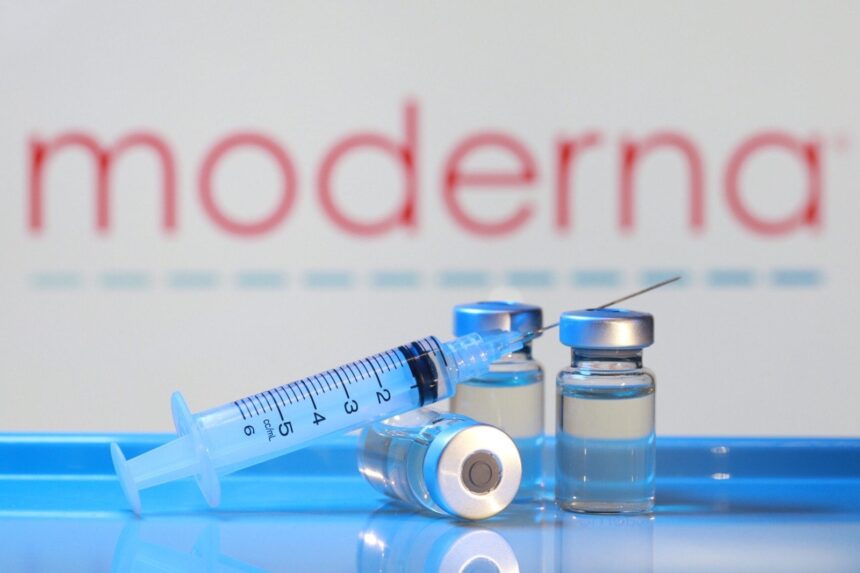
NEW YORK — Moderna has been quietly working on an mRNA vaccine for hantavirus since 2023 in collaboration with South Korea’s VIC-K, raising eyebrows as news of a hantavirus outbreak on the MV Hondius cruise ship continues to circulate.
The preclinical project, which has so far only reached mouse trials with no human data available yet, suddenly gained attention after the cruise ship incident resulted in three deaths and five confirmed infections. Moderna’s stock spiked nearly 10% in the immediate aftermath of the outbreak reports.
The timing has fueled online speculation and debate, with some questioning the coincidence of a vaccine in early development and a real-world outbreak of the same rare virus.
Moderna’s Hantavirus Vaccine Program
According to available information, Moderna partnered with the Korea Vaccine Institute (VIC-K) to develop an mRNA-based vaccine targeting hantavirus. The project remains in the preclinical stage, meaning it has shown promise in laboratory and animal studies but has not yet advanced to human clinical trials.
Hantavirus is a serious zoonotic disease with no approved vaccine or specific antiviral treatment available in the Western world. Severe cases, particularly those involving hantavirus pulmonary syndrome, can have fatality rates ranging from 30% to 40%. The current outbreak on the MV Hondius involves the Andes virus strain, which is one of the few hantaviruses known to allow limited person-to-person transmission under close-contact conditions.
Moderna’s move into hantavirus research fits its broader strategy of expanding its mRNA platform beyond COVID-19 into other infectious diseases with high unmet medical need.
The MV Hondius Outbreak Context
The expedition cruise ship MV Hondius reported the first symptoms in early April during an Antarctica voyage. Three passengers have died, and five others are confirmed infected. The ship is currently en route to Spain’s Canary Islands under strict quarantine after being denied port access elsewhere.
The World Health Organization has confirmed the cases and stated that the risk to the general population remains low, while emphasizing supportive care as the only current treatment option.
The overlap between Moderna’s early-stage vaccine work and the real-world outbreak has sparked intense discussion. Some observers see it as a timely reminder of the value of proactive vaccine research, while others have raised questions about market reactions and prior positioning.
Market Reaction and Investor Interest
Moderna’s stock jumped sharply on the news of the cruise ship cases. While the company has not commented directly on any connection to its vaccine program, investors appear to be pricing in the potential long-term value of having an mRNA platform ready for rare but deadly pathogens.
This reaction mirrors patterns seen during previous health scares, where companies with relevant pipelines saw temporary share price gains based on speculation rather than immediate commercial opportunities.
It is important to note that even if the vaccine candidate proves successful, it would likely take several years to reach full approval and widespread availability.
Broader Questions on Vaccine Development
The situation highlights the complex relationship between public health threats and pharmaceutical innovation. Developing vaccines for rare diseases is expensive and risky, often requiring government or international funding to make economic sense.
Moderna’s early investment in hantavirus research could prove valuable if outbreaks become more frequent due to climate change, increased human encroachment on wildlife habitats, or other factors. At the same time, the rapid market reaction to the cruise ship news underscores how quickly fear can move markets, even when the immediate threat to the general public is considered low.
Health experts continue to stress that hantavirus is primarily transmitted through rodent exposure and does not spread easily between humans in most strains. The current focus remains on containment of the shipboard outbreak and supportive medical care for those affected.
What Comes Next
The MV Hondius is expected to arrive in the Canary Islands soon, where Spanish authorities will conduct thorough screening and monitoring. The WHO and other health agencies are providing technical support.
For Moderna, the hantavirus program remains in early development. Any potential acceleration would depend on further data, regulatory pathways, and funding considerations.
As the story develops, the intersection of a real infectious disease event and ongoing vaccine research is likely to keep the topic in the spotlight. For now, officials continue to emphasize that the risk to the broader public remains low and that proven public health measures, rather than unproven treatments, are the appropriate response.